Alan Dehope (15-ERD-036)
Abstract
High explosives play an integral role in the nation's defense as components in conventional munitions as well as providing an energy source for nuclear weapons. However, the development of higher-performing explosives is limited by conventional organic-based explosives and their formulations. Significant improvement in explosive performance is an unmet scientific challenge that has persisted for decades. High-explosive performance is strongly dependent on material density. Therefore, we postulate that we can realize large gains in detonation performance by exploiting the high densities and large amounts of stored chemical energy available in metal fuels. We intend to prepare energetic ligands (an ion or molecule that binds to a central metal atom) and high-power metal complexes to realize significant gains in detonation performance. We are proposing a path to increase detonation power by extracting the energy of metal oxidation (burning) in a detonation front, with predicted gains greater than 50% over the nitramine explosive CL-20. Such an increase in detonation pressure would have a groundbreaking effect on the field of energetic materials. These performance gains can be realized by leveraging recent organometallic chemistry methodologies that will allow for the creation of a new class of metallic explosives that possess fuel and oxidizer in the same molecule.
We expect to develop an oxidizer-rich organic donor-ligand molecule that will be complexed to a low-valent metal fuel, forming a high-density energetic metal compound. The new material will contain metal fuel and oxidizer in the same molecule and will allow for the oxidation to occur at a very fast rate, resulting in a detonation. The oxidation of the metal fuel will contribute to detonation pressure and velocity. Such a combination is a new concept in energetic materials research, and thermodynamic equilibrium calculations using the Cheetah code for modeling explosives predict significant improvements in detonation performance by burning metal in detonation. Such increases in material performance would enable improvement in weapons systems performance and create new applications for conventional weapons.
Mission Relevance
This research aligns well with the Laboratory's mission in national security, where improvements in weapon system performance are needed. New materials, in support of LLNL's core competency in advanced materials and manufacturing, are required by weapon designers to meet the requirements of smaller, high-performance munitions that will enhance the lethality and bolster the nation's defense, which is relevant to the strategic focus area in stockpile stewardship science.
FY15 Accomplishments and Results
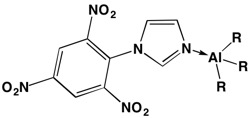
In FY15 we (1) successfully prepared several heterocyclic carbene donor ligands, which contain oxidizing nitro groups (organic compounds containing nitrogen dioxide functional groups), as shown in the figure; (2) characterized the ligands using proton (hydrogen-1) and carbon-13 nuclear magnetic resonance spectroscopy; and (3) worked to determine which of these new ligands are the best candidates to coordinate to metals.