Maxim Shusteff (14-LW-077)
Abstract
Developing effective therapies and countermeasures against viral infections remains extraordinarily challenging and highlights the need for new tools to study viral interactions with host cells. Traditional tissue culture approaches provide investigators with a high degree of experimental control and flexibility, but the static nature of flask-based cell culture significantly distorts infection patterns, evolutionary parameters, and replication dynamics. In contrast, animal studies provide a wealth of information, but the interpretation of results is confounded by the large number of uncontrolled or unknown variables in complex living systems. Bridging technologies are urgently needed to improve upon the unrealistic dynamics in static flask-based tissue cultures, while avoiding the complexity and expense of in vivo experiments. Toward this goal, we have developed a continuous long-term viral culture platform that recapitulates more in vivo dynamics than static culture approaches, while offering greater experimental control. Suspension cells are grown in a bioreactor, and retained using a label-free microfluidic size-separator, while used media and viruses are removed. The same device allows for sampling each of the cell and viral sample streams at user-defined time intervals, providing near-real-time data on experimental state, and population growth and interactions. Using this controllable, automated culture system, multiple cell types have been cultured for over one month, demonstrating flexibility in manipulating population dynamics. To investigate the effect of gradually removing free viruses from the system (akin to the nonspecific clearance of foreign particles by the immune system), we observe changes in the consensus sequence of the variable receptor binding protein gene of the Sindbis virus genome after being cultured in our platform. This platform combines automation and robustness with the flexibility to adjust parameters and add components, opening the door to a variety of high-impact investigations.
Background and Research Objectives
Emerging and re-emerging diseases continue to be a significant threat to public health across all populations, and new tools for virological investigation are urgently needed. The rapid development and dissemination of new molecular tools allows unprecedented interrogation and monitoring of viral genetic sequences over time. However, despite these developments, some standard methods in virology have not advanced, including the standard T-flask-based in vitro tissue culture methods first introduced in the 1950s.
These approaches have given investigators experimental flexibility and a high degree of experimental control to test specific hypotheses. However, the techniques are fundamentally static and serial in nature, requiring manual “passages” of cell and virus populations. This is a re-infection of a new flask after two to five days, when the host cells begin to die off. The limitations of this resource-intensive batch-based methodology leave many unanswered questions about the fundamental mechanisms of viral adaptation and host–virus dynamics. Moreover, little about these in vitro conditions reflects realistic physiological conditions, because the viral load is forced to take on a saw-tooth profile between artificially low and high points, with evolutionary bottlenecks at the low points.1 In contrast, observations in vivo reveal that many viruses (such as hepatitis B and C and HIV) persist for years, mainly because of continual clearance of circulating viral particles from the body by non-specific immune mechanisms and the availability of “naive” uninfected cells.2,3
In contrast, clinical patient data provides the most relevant information, but ethical and logistical constraints preclude preemptive studies, leaving investigators always chasing the most recent outbreaks. In vivo animal models are a valuable platform to conduct experiments with some level of control, yet they can still be overwhelmingly complex, inexact, and expensive. Thus, bridging technologies are desperately needed to address the unrealistic dynamics in static flask-based cultures, and mitigate the complexity and expense of in vivo experiments.
Therefore, the key objective of this work is to capture the dynamic equilibrium that exists in vivo between many viruses and their host cells by building a culturing system that continually clears free virus and re-circulates host cells back to the culture. Using a microfluidic device to accomplish the necessary particle manipulation, this project also tackles the broader challenge of adapting microliter-scale devices for milliliter and larger scale operations. To this end, we have developed a mesoscale engineered system to enable experiments to use automated tissue culture, and demonstrated how it can be used to establish a cell-virus system and probe viral evolution.
Scientific Approach and Accomplishments
In prior work, our team conceptualized and tested a microfluidic acoustic particle separator that uses a “transparent” wall to subdivide the channel, creating asymmetric focusing. The sample to be processed is introduced into the flow-through microdevice, flowing side by side with a clean fluid stream. Particle size-dependent acoustic radiation forces fractionate the sample by selectively transferring large particles to the clean stream, collected from one of the chip outlets. Meanwhile, small particles and soluble species remain in the original solution, and are collected from a separate outlet. This label-free sorting method is particularly appropriate in our application, having been demonstrated as gentle enough to have no effect on cell expression or viability.4–6
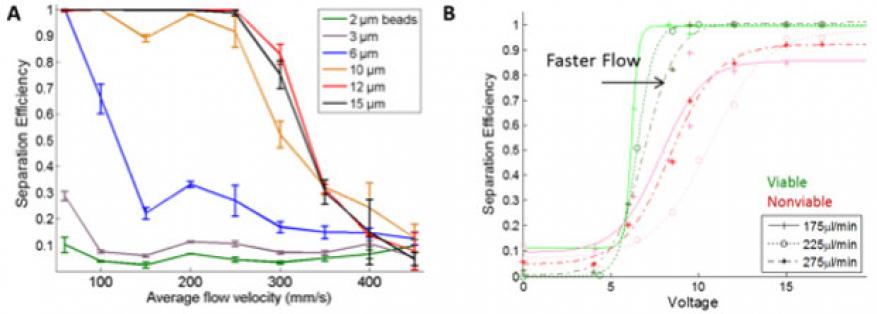
As part of this project, we further advanced this technology, optimizing the devices for high speed separation. Figure 1(A) shows the separation efficiency for microspheres of different sizes at high flow speeds. Significantly, 10-µm beads are extracted from the original stream at 90% efficiency even at 250 mm/s, which is a total flow of 900 µL/min. To our knowledge, this is the greatest linear flow velocity and one of the highest volumetric flow rates among any acoustophoretic devices that have been reported to date. We further demonstrate robust cell-virus separation with three different cell virus pairs: human Raji lymphocytes (5–8 μm diameter) and Dengue virus (50 nm), Raji lymphocytes and Golden Gate virus (est. 100–150 nm), and boa constrictor cells (8–10 µm) and Golden Gate virus. The experiments with Boa cells and Golden Gate virus in particular demonstrated the power of this separation platform, because neither species is well characterized in its physical properties, yet the Livermore acoustic platform achieved the desired separation using only a few small-volume samples. Figure 1(B) shows additional separation efficiency data of viable and non-viable cells, as measured during use of the acoustic device as part of the culture platform developed for this project (see additional discussion below).
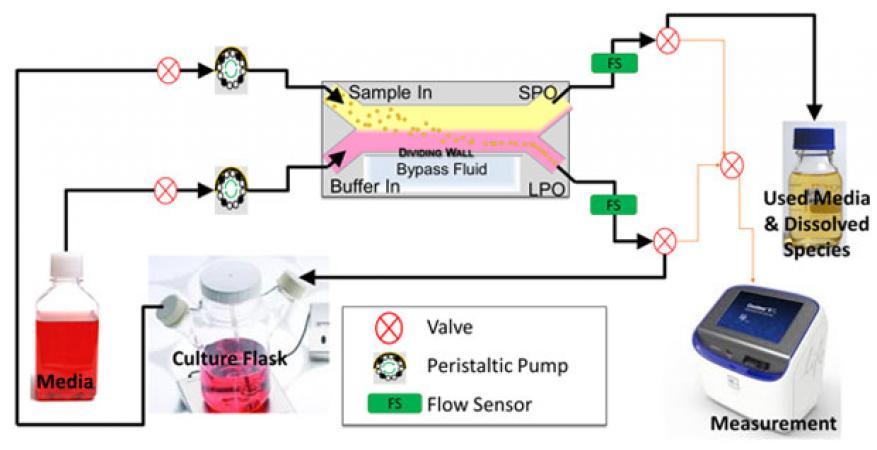
In order to realize a continuous perfused cell culture platform, a system needed to be built that would allow the device to operate continuously and robustly without user intervention, and to process large volumes, because the system runs unattended for extended periods of time. An array of computer-controlled components, including valves, flow sensors, pumps, and sample collectors provides precise control of the system state and operating parameters, with continuous monitoring of relevant variables (Figure 2). Using real-time feedback from flow sensor data, the platform automatically reacts to flow irregularities to resolve common problems and can operate with nearly no operator intervention for weeks at a time. Incorporation of the acoustic separation device not only permits fractionation of cells and viruses by tuning the flow rate and power supplied to the acoustic device, but also provides some ability to differentiate between viable and nonviable cells, as sen in Figure1(B).
To augment the operation of this platform with some predictive power, we developed a mathematical model that estimates changes in populations because of fluidic system inputs and outputs alone (based on flow rates, system configuration, and the separation efficiency given particle sizes). We validated the model by monitoring the concentration of 1 and 10 µm polystyrene beads (because these do not grow or die) over time with the system running under different configurations and operating conditions. Across multi-day tests, microsphere population data matched model predictions reasonably well (Figure 3).
When culturing cells, the microfluidic acoustic size-separation device is used to retain cells in the system while used media with waste products are removed (Figure 2). Using this platform, multiple suspension cell types have been cultured at steady-state conditions (constant cell density) for over one month with limited user intervention. Importantly, the system collects aliquots of the culture at user-defined intervals, enabling near-real-time measurement of growth rates, which become additional parameters in the population model described above. From this information, system parameters can be adjusted to control cell or virus concentration. For example, in Figure 4, THP-1 leukocytic cells7 are maintained at two different steady states, at a high cell density (dark blue), and a low cell density (light blue) by adjusting the rate at which cells are allowed to leave the system (bleed rate). For comparison, the growth curve in a closed static T-flask is shown in red. All three populations display similar initial characteristics, showing exponential growth within the first few days. In comparison to the static flask, in which the cells die rapidly after exhausting the media (negative growth rate), cells in our perfusion system maintain stable growth over time, with the low-density cells maintaining a steady and high growth rate of about 0.6 cells per cell per day.
The possibilities of using this platform to study viral evolution become clear when we use the automated system to establish a cell–virus culture with the Sindbis virus infecting the THP-1 cells. We examined the effect of gradually clearing free virus from the culture, by observing changes in the consensus sequence of the gene coding for the variable receptor binding protein of Sindbis virus. The viral genome was Sanger-sequenced at multiple time points after infection. Analogous control experiments are performed in a spinner flask with no fluid entering or leaving, and a traditional T-25 flask for comparison (Figure 5).
The virus replicated in all three systems, with intriguing sequence changes observed at residue 181, which is part of the viral receptor binding protein, and has been shown to be important in binding to susceptible cells.8,9 Thus, mutations in this residue are expected as the virus adapts to the THP-1 cells, which are different from the Vero cells on which the stock virus was produced. Consequently, two variants appear in most of our samples, corresponding to glutamic acid and lysine at residue 181, while the ancestor virus has only the glutamic acid variant, suggesting that the 181 lysine variant binds more successfully to THP-1 cells. Interestingly, we observe different dynamics between the closed cultures (spinner and static flasks) and the perfused culture. In the closed cultures, a relative increase in the 181 lysine mutant is observed after the first round of viral replication (7 h after infection).10 In contrast, in the perfused culture the relative amount of the glutamic acid mutant increases within the same timeframe. Longer-term experiments with higher-fidelity sequence data (next-generation sequencing) are required to develop a fuller picture and deeper understanding of the mutation mechanisms, which were beyond the scope of the present investigation. However, these preliminary results hint at the possibilities made available by the automated long-term culture platform of measuring and controlling viral evolution parameters.
Impact on Mission
This work has contributed to the Laboratory’s biosecurity mission, creating a new capability for pathogen characterization, studying host and pathogen interactions, and directly impacting the development of countermeasures to biological threats. Insights from studies carried out with this platform are immediately relevant for vaccine production systems operating under similar suspension and perfusion conditions. Moreover, as a novel ex vivo system for understanding human physiology, the work supports one of the core research priorities within the Laboratory’s bioscience and bioengineering core competency. Using this system, it is possible to simulate aspects of the human immune response by growing immune cells in the presence of pathogens, controlling the population of these pathogens, and adding therapeutic compounds or drugs. As such, the technology is synergistic and complementary with other organ-on-a-chip technologies under development at the Laboratory. With this project, a new postdoctoral staff member was hired with expertise in bioengineering. In addition, one of the key project staff completed her doctoral studies with support from the Livermore Graduate Scholar Program, and at the completion of her doctorate, was hired as an Livermore postdoctoral researcher.
Conclusion
This project successfully developed a continuous automated long-term culture platform that minimizes un-physiological disruptions of cell and viral populations, removes the need for manual attendance of the culture, and offers aspects of experimental control unavailable with traditional culture systems. Overall, we have demonstrated the ability to use a microfluidic device to monitor and control large-scale cell population behavior. As a first demonstration of probing viral evolution dynamics, we examined changes in the Sindbis virus consensus sequence resulting from being grown in our platform. The technology is poised to yield a wealth of new insights when coupled with next-generation sequencing methods for high-fidelity genetic sequence analysis. Such work can also provide the necessary data for computational analysis and predictive modeling of viral evolution dynamics, a major goal of the Defense Advanced Research Projects Agency’s INTERfering and Co-Evolving Prevention and Therapy program aimed at developing transmissible, engineered antiviral therapeutics. More broadly, we expect this platform to bring forth revolutionary studies on infection dynamics and pathogen evolution. We developed productive collaborations with experts in the virology community, both internally and externally. Of particular note is the Weinberger laboratory of the Gladstone Institute for Virology and Immunology at the University of California, San Francisco, experts in developing HIV therapeutics, with whom we have co-authored two journal articles. This project has also attracted interest from two commercial entities.
References
- Gummuluru, S., C. M. Kinsey, and M. Emerman, “An in vitro rapid-turnover assay for human immunodeficiency virus type 1 replication selects for cell-to-cell spread of virus.” Virology 74(23), 10882 (2000).
- Nowak, M. A., and R. May, Virus dynamics: mathematical principles of immunology and virology. Oxford University Press, New York, NY (2001).
- Perelson, A. S., and P. W. Nelson, “Mathematical analysis of HIV-1 dynamics in vivo.” SIAM Rev. 41(1), 3 (1999).
- Burguillos, M. A., et al., “Microchannel acoustophoresis does not impact survival or function of microglia, leukocytes or tumor cells.” PloS One 8(5), e64233 (2013).
- Hultström, J., et al., “Proliferation and viability of adherent cells manipulated by standing-wave ultrasound in a microfluidic chip.” Ultrasound Med. Biol. 33(1), 145 (2007).
- Wiklund, M., “Acoustofluidics 12: Biocompatibility and cell viability in microfluidic acoustic resonators.” Lab Chip 12(11), 2018 (2012).
- Tsuchiya, S., et al., “Establishment and characterization of a human acute monocytic leukemia cell line (THP-1).” Cancer 26(2), 171 (1980).
- Lustig, S., et al.,"Molecular basis of Sindbis virus neurovirulence in mice." Virology 62(7), 2329 (1988).
- Navaratnarajah, C. K., and R. J. Kuhn, “Functional characterization of the Sindbis virus E2 glycoprotein by transposon linker-insertion mutagenesis.” Virology 363(1), 134 (2007).
- Frolov, I., et al., “Early events in alphavirus replication determine the outcome of infection.” Virology 86(9), 5055 (2012).
Publications and Presentations
- Bora, M., et al. “Efficient coupling of acoustic modes in microfluidic channel devices.” Lab Chip 15, 3192 (2015). LLNL-JRNL-664577. http://dx.doi.org/10.1039/c5lc00343a
- Fong, J., et al, “A microfluidic platform for precision small-volume sample processing and its use to size separate biological particles with an acoustic microdevice.” JoVE e53051 (2015). LLNL-JRNL-665235. http://dx.doi.org/10.3791/53051
- Fong, E. J., et al., “Acoustic focusing with engineered node locations for high-performance microfluidic particle separation.” Analyst 139(5), 1192 (2014). LLNL-JRNL-640796.
- Fong, J., et al., Autonomous perfusion platform to manipulate viral evolution. 2016 American Society for Microbiology Biodefense and Emerging Diseases Research Mtg., Arlington, VA, Feb. 8–10, 2016. LLNL-ABS-678968.
- Fong, E. J., et al., Continuously variable node position in a high-throughput acoustofluidic device. Micro Total Analysis Systems Conf., San Antonio, TX, Oct. 26–30, 2014. LLNL-PROC-657658.
- Fong, E., et al., Manipulating viral evolution: An automated perfusion system. SLAS 2016 5th Annual Intl. Conf. and Exhibition, San Diego, CA, Jan. 23–27, 2016. LLNL-POST-680927.
- Fong, J., et al., Smart automated platform for precise manipulation of population dynamics. (2016). LLNL-PRES-677687.
- Jung, S. Y., et al., “Spatial tuning of acoustofluidic pressure nodes by altering net sonic velocity enables high-throughput, efficient cell sorting.” Lab Chip 15, 1000 (2015). LLNL-JRNL-665514. http://dx.doi.org/10.1039/c4lc01342e