Vanessa Tolosa (16-FS-013)
Project Description
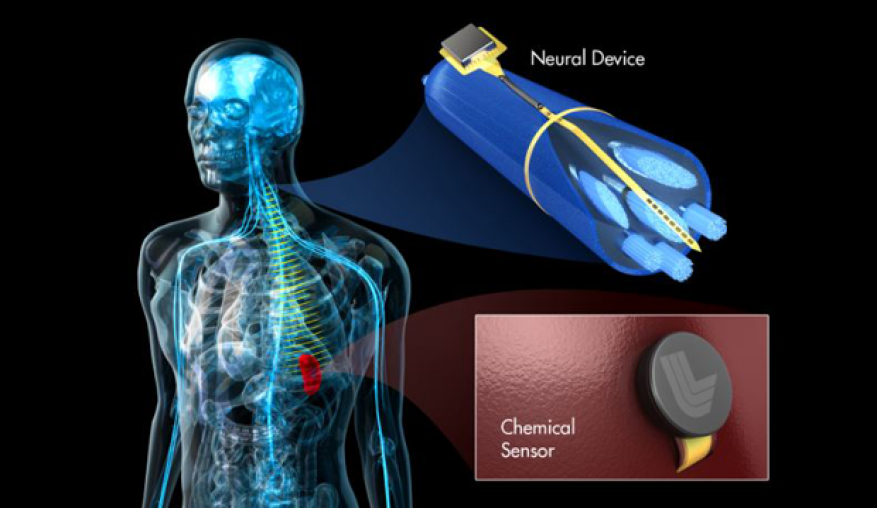
In 2015, University of Virginia scientists announced their finding of a direct link between the brain and the immune system. Their discovery of lymphatic vessels in the mouse brain has changed the long-standing view that the body's immune cells, carried by lymphatic vessels, don't interact with the brain. This connection opens important new questions about how brain functions are affected by the body's immune system. The news comes as the case for another link, one between the brain and the gut's microbiota, gains attention in the scientific community. Clinical studies have shown that vagus nerve stimulation has the power to affect a host of diseases and disorders, from arthritis to migraines. The vagus cranial nerve plays a key role in the functioning of the heart and digestive tract. Vagus nerve stimulation as a treatment, however, has not gained widespread use because the mechanism by which it works is largely unknown and its effects are difficult to reproduce. We plan to investigate the feasibility of using peripheral nerve stimulation on diseases and disorders by developing neural interfaces to interact with both systems simultaneously. For this feasibility study, we will focus on developing interfaces for the vagus nerve and gastrointestinal system. In doing so, we will leverage Livermore's expertise in developing flexible, implantable device technologies.
We intend to investigate the feasibility of uncovering some of the most fundamental biological mechanisms underlying the gut–brain link, and how it may be used to develop more effective therapies for brain, gut, and immune system disorders. We will leverage Livermore's expertise in developing and fabricating biologically compatible implantable medical devices for modulation and recording of the body's electrical activity. Currently, the devices are used to modulate the retina and to study the central nervous system. In this project, we will expand this neural-interface platform to interface with the peripheral nervous system and organs. We will design a system that can electrically stimulate the vagus nerve while simultaneously recording the electrical and chemical response of the gut. The platform can be further expanded to study the immune system and central nervous system simultaneously. A successful outcome will enable scientific discoveries and clinical therapies that are currently limited by available technologies. Our feasibility study may enable researchers to investigate the body's ability to heal itself through peripheral nerve modulation. With a better understanding of the fundamental mechanisms behind the gut–brain interactions and peripheral nerve stimulation, clinicians will have new tools for personalized medicine.
Mission Relevance
Our project supports the Laboratory's strategic focus area of chemical and biological security, a priority of which is developing tools for rapid development of medical countermeasures. The Laboratory's strengths in microscopic fabrication of biological interfaces and material science engineering will be used to develop interfaces that are biologically compatible and not yet commercially available, which is aligned with the core competency of bioscience and bioengineering. Additionally, the project will nurture collaborations with industry and academia.