Timothy Rose (16-ERD-008)
Executive Summary
Developing a science-based experimental understanding of the correlation between nuclear fireball conditions and debris chemistry builds confidence in existing fractionation correction methods and could identify needed improvements, enhancing the Laboratory's core competency in nuclear weapons science and supporting DOE and NNSA goals in nuclear security and addressing emergency response and the threat of nuclear terrorism.
Project Description
Post-detonation nuclear debris is the product of extreme chemical fractionation and mixing processes between the nuclear device and background material. These processes alter the chemistry of the nuclear debris so that it no longer reflects the chemistry of the source weapon. Radiochemical measurements of the debris must be corrected for these chemical distortions before the data can be used to draw inferences regarding the characteristics of the nuclear device. Empirical data correction methods that were developed during nuclear testing are still being used today. These methods are reliable, but offer no insight into the physical mechanisms controlling chemical fractionation. Our objective is to investigate chemical fractionation processes under conditions relevant to nuclear fireball condensation. This work will advance our experimental and theoretical knowledge of plasma condensation chemistry and is needed to build a science-based understanding of radiochemical correction methods. We will measure correlations between the physical and chemical properties of condensing plasmas at temperatures less than 1 eV using a steady-state inductively coupled plasma-flow reactor suitable for studying high-temperature chemistry in partially ionized plasmas. The flow rate of the system is adjustable, enabling the simulation of different cooling and mixing histories. This allows us to examine the interplay between cooling time scales and chemical fractionation patterns, and may offer insights into the relative roles of thermodynamic versus kinetic mechanisms in determining chemical partitioning. Comparison of the experimental results with data from historic nuclear tests will establish a critical link between the experimentally calibrated observables and the fractionation patterns in actual debris.
We will determine empirical fractionation ratios for pairs of chemical elements as a function of cooling and mixing rates, matrix composition, and oxygen fugacity (the pressure needed at a given temperature to enable properties of a nonideal gas to satisfy the equation for an ideal gas). The sensitivity of chemical fractionation to these factors will be explored by varying each parameter over a range in values relevant to fireball condensation. Constraining uranium volatility is the top priority. This work will result in the first data set of its kind that directly relates fallout fractionation patterns to measured physical phenomena. The results will provide a basis for developing advanced computational models to simulate chemical fractionation using chemical kinetics or molecular dynamics codes. These models will, in turn, inform the development of predictive models for chemical fractionation patterns in fallout.
Mission Relevance
Developing a science-based experimental understanding of the correlation between fireball conditions and debris chemistry builds confidence in existing fractionation correction methods and may identify needed improvements, enhancing the Laboratory's core competency in nuclear weapons science and supporting DOE and NNSA goals in nuclear security and addressing emergency response and the threat of nuclear terrorism. This work will leverage our broad knowledge of radiochemical distributions in debris, further supporting Livermore's core competency in forensic science.
FY17 Accomplishments and Results
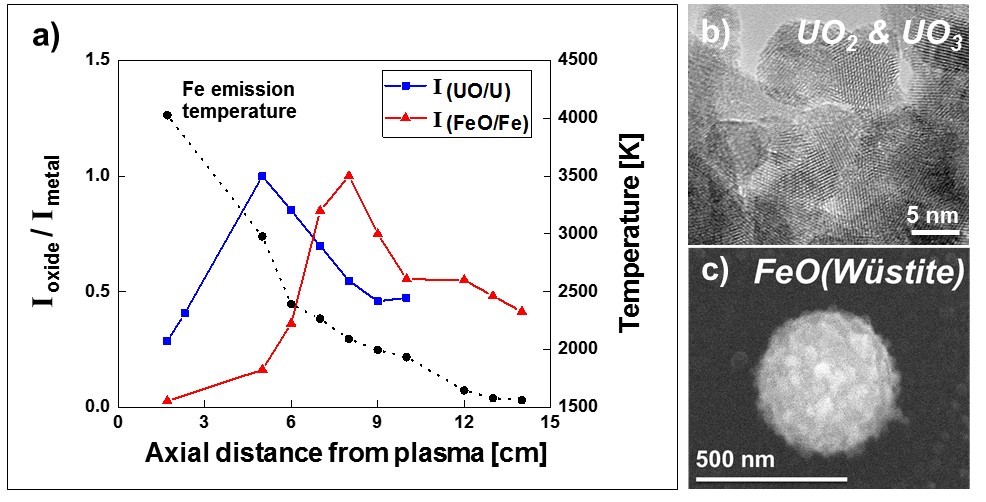
In FY17 we (1) performed particle characterization using scanning and transmission electron microscopy and developed emission spectra diagnostics; (2) determined that for experiments in the iron–aluminum–oxygen system, iron–oxygen and aluminum–oxygen dimer chemical structures show distinct condensation temperatures (from emission spectra) that correlate with relative abundances of iron and aluminum oxide particles collected in different temperature regimes, which is consistent with chemical fractionation; (3) performed experiments on uranium–oxygen and uranium–iron–oxygen, although behind schedule because of delays in system engineering and repair of our pulsed infrared laser system; and (4) began tests using reducing agents to modify oxygen conditions.
Publications and Presentations
Koroglu, B., et al. 2017. "Gas phase chemical evolution of uranium and iron oxides." Presentation at Lawrence Livermore National Laboratory, Livermore, CA. LLNL-POST-739560.
———. 2017. "Plasma flow reactor for steady state monitoring of physical and chemical processes at high temperatures." Review of Scientific Instruments 88, no. 9: 093506. doi:10.1063/1.5001346. LLNL-JRNL-730618.
———. 2017. "Preliminary experimental results using a steady state ICP flow reactor to investigate condensation chemistry for nuclear forensics." Presentation at Lawrence Livermore National Laboratory, Livermore, CA. LLNL-PRES-707819.
Rose, T. et al. 2017. "Experimental studies of chemical fractionation." Presentation at Lawrence Livermore National Laboratory, Livermore, CA. LLNL-PRES-716637.
———. 2017. "Why nuclear forensics needs new plasma chemistry data." Presentation at Lawrence Livermore National Laboratory, Livermore, CA. LLNL-PRES-707684.