Anna Belle | 16-ERD-035
Overview
Biological systems communicate and function through a series of complex biochemical and electrical interactions. The ability to monitor these diverse interactions in real time and in physiologically relevant settings can be a powerful tool in determining the biological causes of diseases and uncovering the mechanisms underlying the body's response to chemical agents. While there are abundant electrical recording instruments, there are few chemical recording platforms. This is despite the fact that only a fraction of the body's signaling mechanism is electrical and the majority is biochemical. Over 500 quadrillion chemical reactions occur in our bodies each day. The goal of this project was to provide a fundamental understanding of the body's response to bio-agents and drugs by developing an advanced chemical-sensor platform. Individual sensors monitored cell health by measuring multiple biomarkers simultaneously, in real time. We first created a sensor array and the recording instruments needed to build this advanced sensor platform. Then we addressed the problems of sensor lifetime by using a novel technique of chemically engineering enzymes with nanometer-scale complexes that improve enzyme stability. To improve sensor sensitivity, we custom engineered strings of nucleotides to specifically recognize complementary strings of nucleotides on our target protein of interest and also increased the electrode sensing interface with a custom roughening technique. Finally, we enhanced the fabrication of electrode arrays by incorporating carbon-based materials into our micro-fabricated array platform, thereby opening the possibilities for new sensor methods on our platform.
Background and Research Objectives
New diagnostic tools that can monitor the functionality and health of the human body are required to understand and counter many of the major health-related burdens on our country today from mental illness and neurodegenerative disorders to sepsis and heart disease. Better metrics for detecting and treating the causes and symptoms of neurological and other organ disorders are needed but depend on the development of tools that can provide them. The goal of our project was to provide a fundamental understanding of the body's response to bio-agents and drugs by developing an advanced chemical-sensor platform whereby individual sensors would monitor cell health by measuring multiple biomarkers simultaneously, in real time.
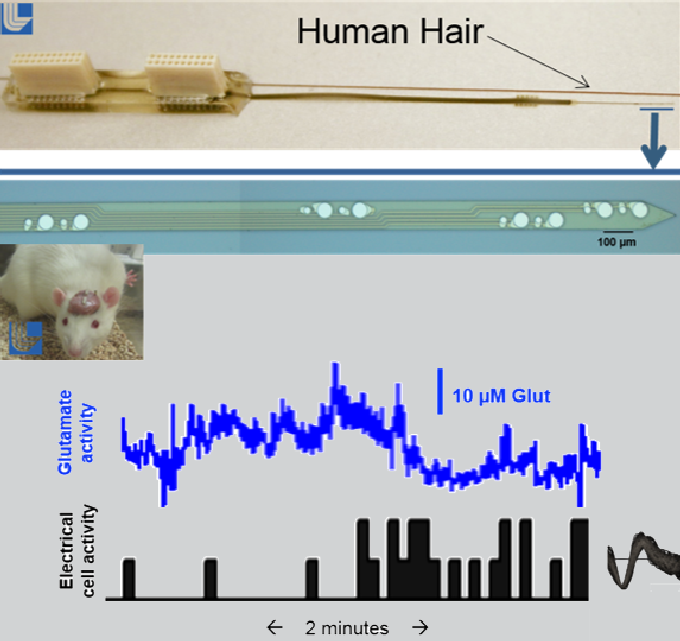
Our initial work focused on implementing multimode sensing. We created the devices and integrated four unique pieces of electrochemical equipment to enable these systems to collect data from multiple sensor types simultaneously. Our first step toward this goal was the creation and onsite animal testing of the first-ever flexible 16-sensor array that collects glutamate and electrical activity in the brain simultaneously (see figure). From this initial study we quickly learned that our microsensors needed enhanced chemical sensitivity and an extended lifetime in vivo to answer many of the pressing questions about how the brain works. We focused our efforts on improving the performance of our chemical sensors using established multimode sensing technology.
Multimodal probe ( top ) capable of collecting simultaneous electrical and chemical data ( bottom ) in the brain of rodents. Top: Probe contains an array of 16 electrodes that can monitor electrical (smaller 20-µm electrodes) and glutamate (larger 50-µm electrodes) in four brain regions simultaneously. Bottom: Glutamate and electrical activity sensed from two adjacent sensors while the animal was moving around his cage.
Impact on Mission
Our chemical sensors are a rapid nondestructive method for monitoring cell health that enables development of countermeasures against biological and chemical threats in support of Lawrence Livermore National Laboratory's strategic focus area in chemical and biological security. Furthermore, the sensors can be modified to enable electrochemical detection both in a clinic and in the field. The biosensors developed on this project are a new capability within the Laboratory's Center for Bioengineering that have attracted the interest of new external collaborators and enabled the Laboratory to maintain its position at the leading edge of this field.
Conclusion
Here we advanced multimode sensors to provide a fundamental understanding of the body's response to bio-agents and drugs. The next steps include transitioning this research to external funding sources to continue to combine sensing modalities on a single probe. We are actively pursuing funding opportunities both independently and in conjunction with external collaborators (users of our sensors).
Publications and Presentations
Belle, A.M., et al. 2016. "Implantable, Flexible Probes for Electrical Stimulation and Recording of Unit Activity and Chemical Release." Monitoring Molecules in Neuroscience Meeting, Gothenburg, Sweden, June 2016. LLNL-POST-692818.
——— . 2017. "Flexible Microelectrode Arrays for Monitoring and Manipulating Neuronal Dynamics." Pittsburg Analytical Chemistry Conference, Chicago, IL, March 2017. LLNL-PRES-725690.
——— . 2017. "Nanoscale Roughened Thin Film Electrodes for Flexible Neural Probe and Bio-Sensing Applications." 62nd International Conference on Electron, Ion, and Photon Beam Technology and Nanofabrication, Orlando, FL, May 2017. LLNL-PRES-732168.
——— . 2018. "New Technologies for Rapid Monitoring of Neurochemicals In Vivo." Pittsburg Analytical Chemistry Conference, Orlando, FL, March 2018. LLNL-PRES-746609.
——— . 2018. "Nanoscale Toughened Flexible Neural Probe for Chronic Biosensing Applications." Meeting of Materials Research Society, Phoenix, AZ, April 2018. LLNL-PRES-746609.
——— . 2018. "A Longer Lasting Biosensor Array: Improving and Extending Performance of Enzymatic Electrochemical Sensors." 29 th World Conference on Biosensors, Miami, FL, June 2018. LLNL-POST-752639.
Ivanovskaya, A. N., et al. 2018. "Electrochemical Roughening of Thin-Film Platinum for Neural Probe Arrays and Biosensing Applications." Journal of The Electrochemical Society 2018, 165 (12), G3125―G3132. doi: 10.1149/2.0221812jes. LLNL-JRNL-746398.
Yorita, A.M., et al. 2017. "Prolonging the Lifetime of Enzymatic Biosensors for Neurotransmitter Detection." Meeting of the Society for Neuroscience, Washington, DC, November 2017. LLNL-POST-752546.
——— . 2018. "Prolonging the Lifetime of Enzymatic Biosensors for Neurotransmitter Detection." Pittsburg Analytical Chemistry Conference, Orlando, FL, March 2018. LLNL-PRES-746934.
——— . 2018. "Fabrication and Characterization of More Robust and Multiplexed Biosensors Using Flexible Polymer Microelectrode Arrays." Meeting of the Society for Neuroscience, San Diego, CA, November 2018. LLNL-POST-760796.