Fang Qian (16-FS-026)
Project Description
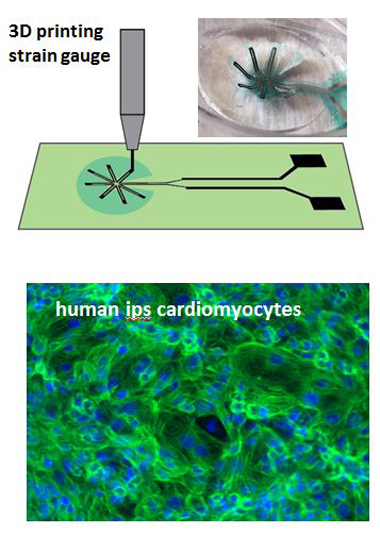
Organ-on-chip devices allow researchers to evaluate complicated and interacting effects of genotype, disease state, and drug intervention on organ function with high throughput. For heart-on-chip research, measuring the contractile force of the tissues is essential. The most robust current design is the “pillar” platform. As heart cells contract, they displace polymer microscopic pillars, exerting a force that is proportional to the pillar's displacement. This platform has limitations that reduce its usefulness as a research tool, including a bulky camera setup, the system's two-dimensional measurement missing subtleties of heart tissue's three-dimensional operation, and nonlinearity in pillar bending that reduces accuracy. We plan to assess the feasibility of a novel, proposed "peacock" device (see figure) that measures cardiac force on a chip with potentially greater accuracy and more fidelity to the three-dimensional structure of heart tissues. It is an electrical device consisting of flexible polymer cantilever beams arranged radially (similar to a peacock's feathered tail) in a network with a microscopic strain gauge deposited radially on the cantilever beams. The beams are interconnected by thin lines of polymer, forming a mesh-like geometry, to improve mechanical stability and ensure uniform force distribution. After seeding onto the peacock device, cardiac cells anchor on the polymer cantilever beams. Cell contraction causes the polymer cantilever to bend upward, which can be electrically recorded by the strain gauge, and the contraction force can be calculated. We will employ micro-fabrication methods to develop proof-of-concept peacock devices, and test their electromechanical response with cardiac tissues grown on the platform.
We expect to determine the feasibility of fabricating peacock devices for cardiac force measurements through multistep photolithography and metal deposition that will feature tunable size, an engineered capability to bend upward, and functional electrical sensors that respond to strain. The peacock arrangement results in an extraordinarily flexible material and improved device sensitivity. In vitro cultured cardiac tissue generates force that is very small and difficult to measure. Despite the use of a stiff polymer, the device's flexibility is possible because the material is thin (<1 µm) and also meshed. We will attempt to record electrical resistance change associated with cardiac beating, and use this as a quantitative force indicator. We intend to address possible technical issues and generate preliminary experimental data, based on which we will be able to provide a valid evaluation of the feasibility, potential, and risk of the peacock platform. If successful, our device will represent a new force-sensing platform class with novel functionality suited specifically to the heart-on-chip technology. The new tool will allow robust and simultaneous electrical and mechanical measurements to be obtained with high throughput. Effects of genotype, disease state, and response to single or multiple drugs on these essential functions could open the door to novel treatment and prevention strategies for people struggling with cardiovascular disease, and those predisposed to it.
Mission Relevance
Our determination of the feasibility of microscale fabrication of a peacock device for measuring cardiac force supports Livermore's core competency in bioscience and bioengineering. The project will employ the Laboratory's strengths in microscopic fabrication of biological interfaces and materials science engineering to develop interfaces that are biologically compatible and fabricated in ways not commercially available. Additionally, the project will nurture collaborations with external research institutions.
FY16 Accomplishments and Results
In FY16 we (1) successfully fabricated the peacock device, by combining conventional micro-fabrication and advanced three-dimensional printing techniques; (2) determined that the device met the key requirements of having flexible cantilever beams that can bend upward by small forces, a radial micro-strain gauge deposited on the beams that is highly electrically conductive, and respond electrically to the beams bending upwards; and (3) established our cardiac lab and began the first cardiac culture. Both rat and human cardiac cells derived from stem cells have been cultured on plates and on peacock devices.